Mutations in all these genes cause Noonan syndrome or Noonan-like phenotypes. Using immunoprecipitation of endogenous LZTR1 ( 600574) followed by Western blotting, Umeki et al. MRAS also functions as a targeting subunit, as membrane localization is required for efficient RAF dephosphorylation and ERK (see 601795) pathway regulation in cells. (2018) showed that MRAS and SHOC2 function as PP1 regulatory subunits, providing the complex with striking specificity against RAF. MRAS ( 608435), SHOC2, and protein phosphatase-1 (PP75) interact to form a heterotrimeric holoenzyme that dephosphorylates the S259 inhibitory site on RAF kinases, activating downstream signaling. 
(1998) cloned a structural and functional SUR8 homolog in humans that specifically binds K-Ras ( 190070) and N-Ras ( 164790) but not H-Ras ( 190020) in vitro. By use of EST primers and 5-prime RACE, Sieburth et al. The sur8 protein interacts directly with Ras but not with the Ras(P34G) mutant protein, suggesting that sur8 may mediate its effects through Ras binding. 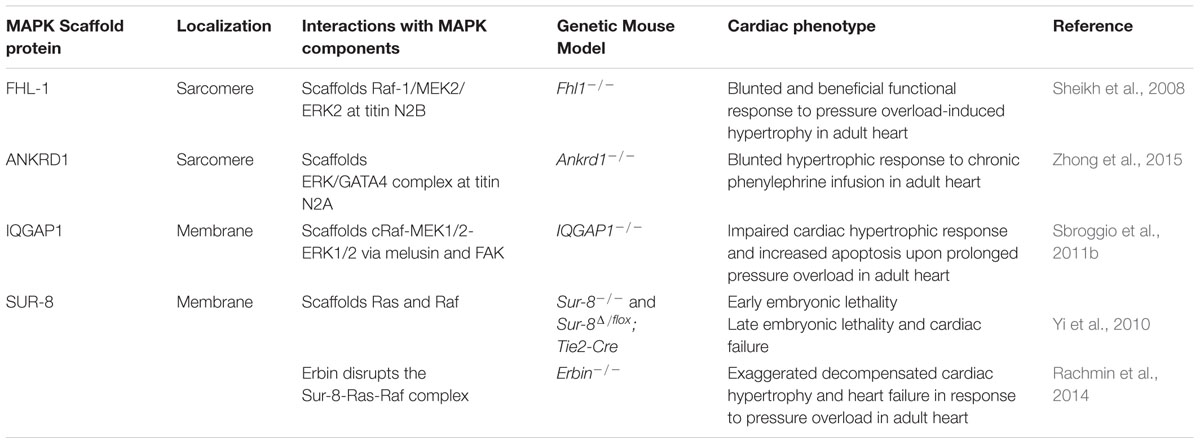
Sur8 encodes a conserved protein that is composed predominantly of leucine-rich repeats. Sur8 appears to act downstream of or in parallel to Ras but upstream of Raf. The authors found that reduction of sur8 function suppresses an activated Ras mutation and dramatically enhances phenotypes of mpk1/sur1 MAP kinase (see 176948) and ksr1 ( 601132) mutations, whereas increase of sur8 dosage enhances an activated Ras mutation. elegans, which positively regulates Ras-mediated signal transduction during vulval development. (1998) identified and characterized the sur8 gene in C. Among fetal tissues, brain showed the highest expression. The authors noted that expression of SHOC2 was 6 times higher in polymorphonuclear (PMN) leukocytes than in mononuclear cells, and suggested that SHOC2 might be important to proliferation or survival of PMNs. In the adult human cDNA panel, the highest expression was observed in testis, with relatively high expression in several immune tissues, including spleen, bone marrow, tonsil, and lymph nodes. (2010) examined the relative expression of SHOC2 in various tissues, including blood leukocytes and lymphocytes. (1998) concluded that this genetic system identified a conserved gene implicated in mediating FGF receptor signaling in C. However, phosphorylation of these residues was not required for soc2 function in vivo, and SHOC2 was not observed to be tyrosine phosphorylated in response to FGF stimulation. (1998) showed that within the leucine-rich repeats of both soc2 and SHOC2 are 2 YXNX motifs that are potential tyrosine-phosphorylated docking sites for the SEM5/GRB2 Src homology 2 domain. They showed that SHOC2 mRNA was expressed in all tissues assayed and that the SHOC2 protein is localized to the cytoplasm. They identified a putative human homolog, SHOC2, which is 54% identical to soc2. (1998) showed that soc2 encodes a protein composed almost entirely of leucine-rich repeats, a domain implicated in protein-protein interactions. Two of these genes were soc1 and soc2, symbolized thus for 'suppressor of clear (Clr)' phenotype the third was sem5. In this organism, they screened for genes that suppress the activity of an activated form of the EGL-15 FGF receptor consistent with the functioning of these genes downstream of EGL-15. (1998) shed light on the intracellular signaling pathways that mediate these processes by studies in Caenorhabditis elegans. Activation of fibroblast growth factor (FGF) receptors elicits diverse cellular responses, including growth, mitogenesis, migration, and differentiation. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
